 |
|
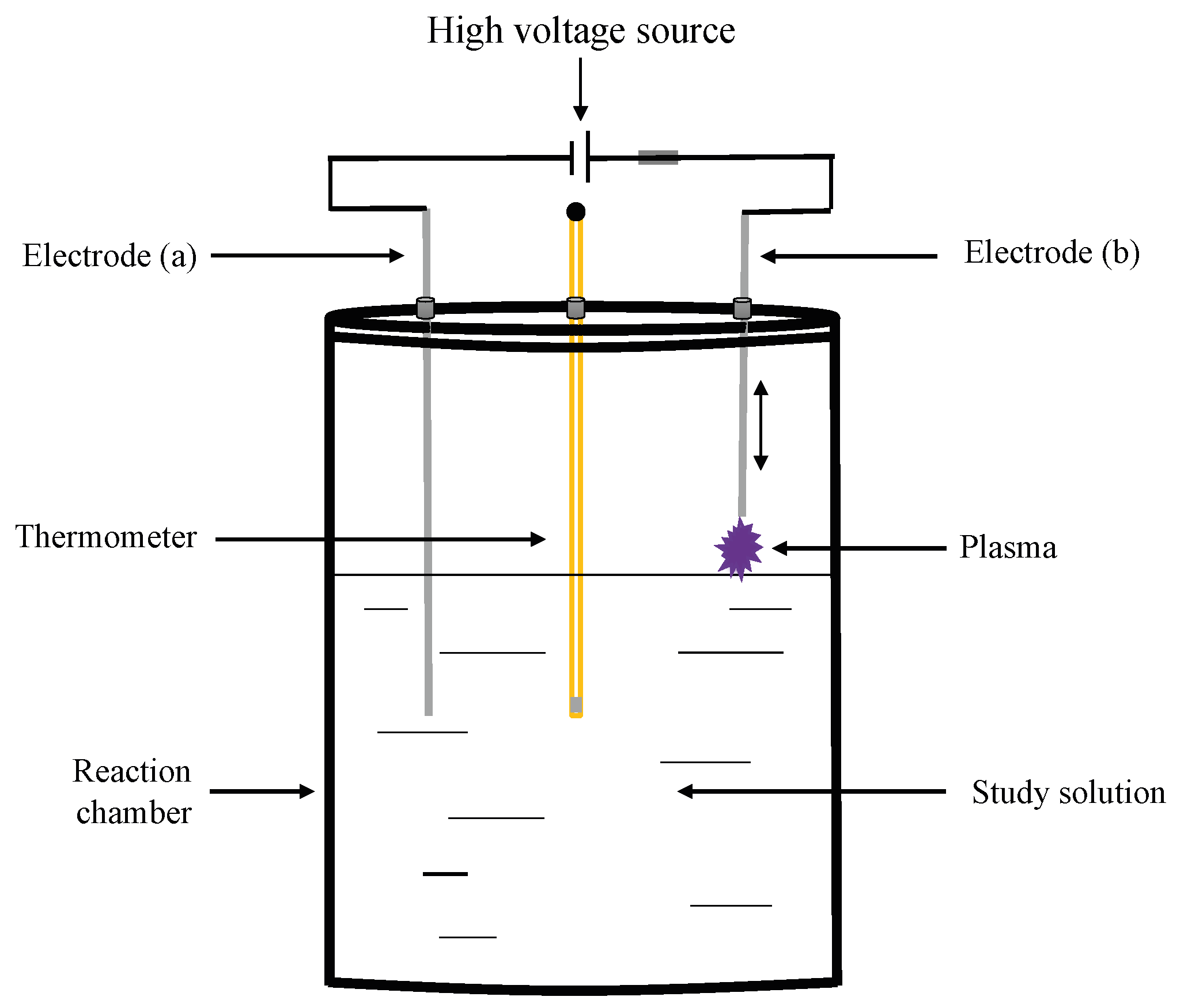
The degradation of Acid Black 52 (AB52) dye in a liquid medium derived from exposure to a non-thermal plasma of air at atmospheric pressure is quantified experimentally. The changes observed in the dilution are measured and compared as a function of the electrical power of plasma generation (15.0 W, 22.5 W, 30.0 W, and 45.0 W), as well as the exposure time (two hundred minutes per electrical power). Dye degradation for each electrical power of plasma was established by comparing the results from UV / VIS spectrophotometry measurements (absorbance, concentration, and percentage of degradation), pH, COD, and TOC. In addition, the toxicity change dilution was determined by performing bioassays using {\it Lactuca sativa} lettuce seeds. The changes for each of the variables measured as a function of the electrical power of plasma generation show the following specific mathematical behaviors: Logarithmic (Temperature, TOC, and pH), potential (COD), and linear (Absorbance). Obtained results showed that the increase in the degradation of the dye and the change in toxicity depends directly on the electrical power and the time of exposure to plasma. The maximum degradation obtained from the samples analyzed at the end of the experiments corresponds to 45.0 W, that is, higher electrical power, a higher percentage of degradation.
Keywords: non-thermal plasma, water treatment, advanced oxidation processes, toxicity, degradation.
|
|
 |

